Description
Safeko Nitrile Exam Glove
Chemotherapy Drug Tested Nitrile Powder Free and Latex Free Examination Gloves are an important component of every hospital safety program and are used by a large number of hospital staff. Hospitals prefer to standardize on one glove that meets the specifications of the Safeko Nitrile Exam Glove with Chemo Drug testing so that there is no staff confusion about which glove to select.
SPH Medical provides both short and long term supplies for hospitals delivering Nitrile Gloves to ensure that clinical staff are properly protected.
The SPH Medical gloves are manufactured under the Safeko brand with strict quality control in two industry leading factories, one located in Malaysia, and another in China. Factory diversity improves the strength of our supply chain and ensures that we can deliver essential supplies to our customers.
Safeko Nitrile Exam Grade Gloves are Chemo Drug Tested. They are available in sizes Small through XL with XXL available as a special order.
- Color: Blue
- Material: Nitrile
- Sizes: Small through Extra Large with XXL provided upon special order
- Packaging: Gloves are sold in boxes of 100 and Cases of 10 boxes = 1000 gloves
- Grade: Exam Grade with AQL 1.5 quality control, Chemotherapy Drug Tested
- Latex: 100% Latex FREE
- Powdered: Powder FREE
- Textured: Textured fingers only
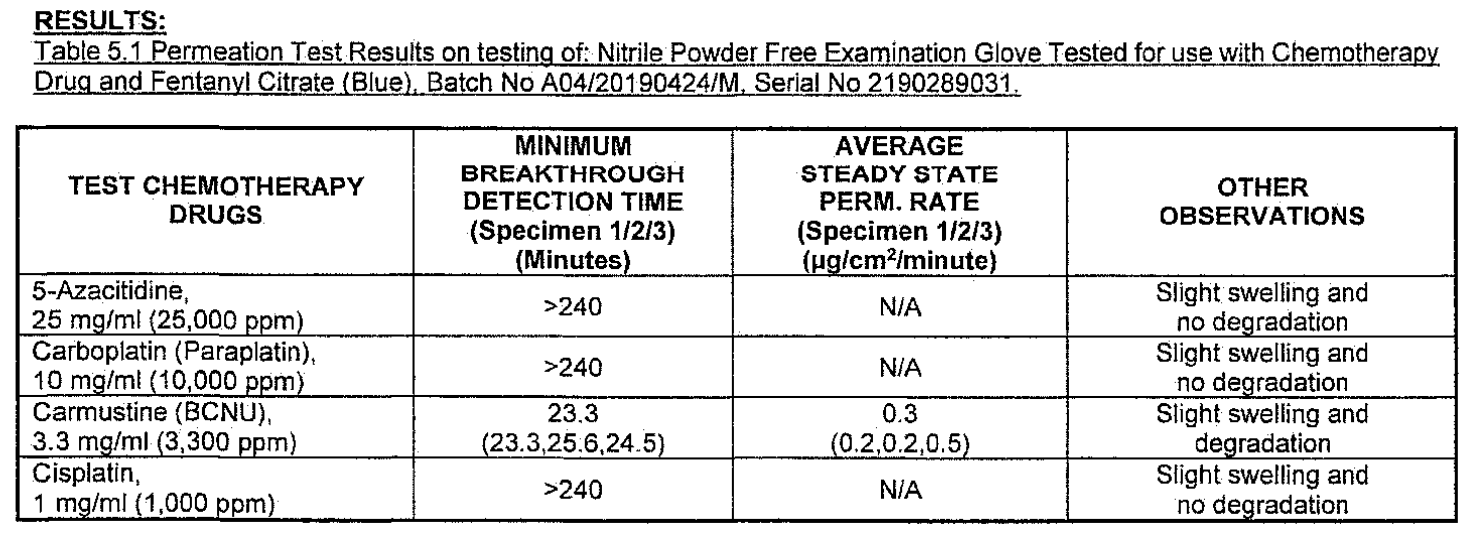
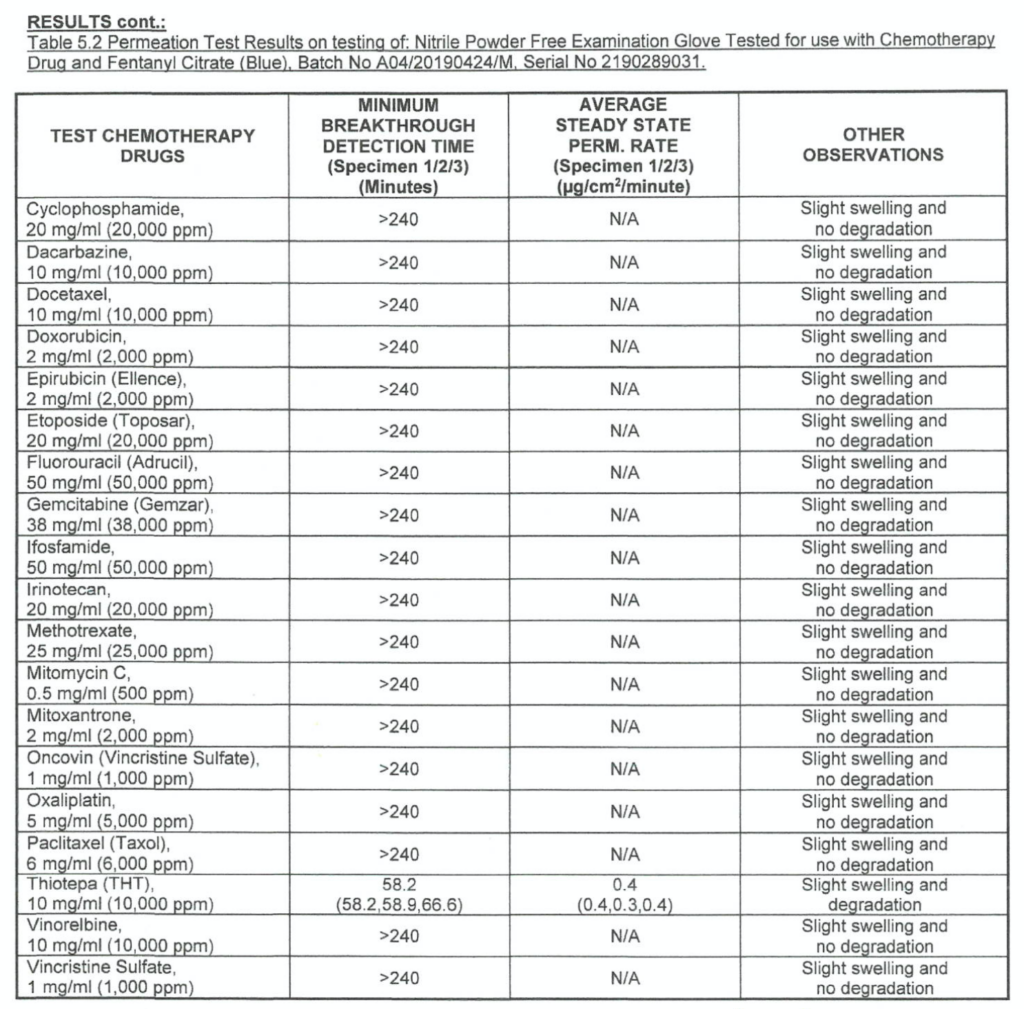
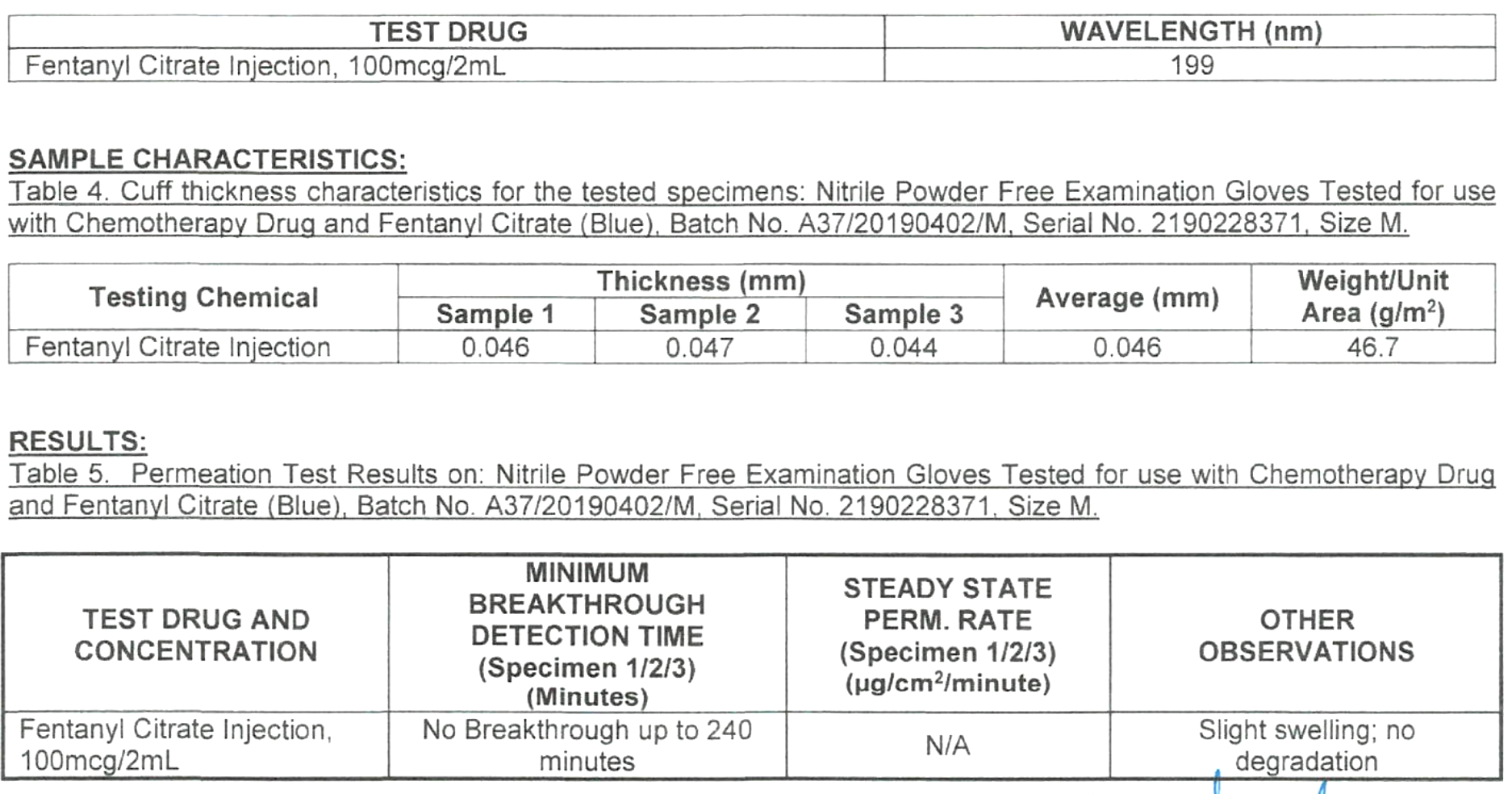
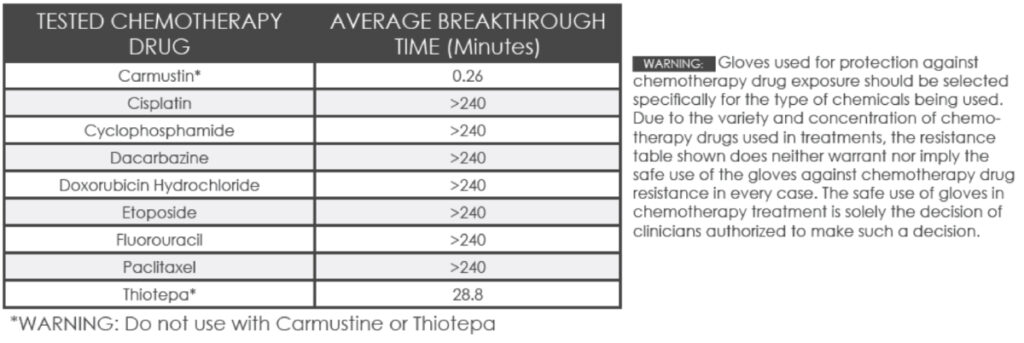
- Chemo Rated: Chemotherapy Drug Tested – see charts below.
- Rating for SK-1009: ASTM F1671, FDA 21 CFR177
- Rating for SK-1038: FDA 21 CFR 177.26
Product Codes are as follows:
- SK-1009-BL – Malaysia Factory
- 6010 Small Gloves, Glove Weight is 2.8g
- 6011 Medium Gloves, Glove Weight is 3.0g
- 6012 Large Gloves, Glove Weight is 3.5g
- 6013 XL Gloves, Glove Weight is 4.0g
- SK-1038 – China Factory
- 6138-S Small Gloves, Glove Weight is 3.1g
- 6138-M Medium Gloves, Glove Weight is 3.5g
- 6138-L Large Gloves, Glove Weight is 3.7g
- 6138-XL XL Gloves, Glove Weight is 3.9g
What is AQL 1.5?
AQL means Acceptable Quality Limit. This strict quality control standard ensures that there is a limit to the number of defects that are acceptable when in a random selected portion of a product batch.