Description
Contact SPH Medical today to Get a Quote and begin saving on your Nitrile Exam Gloves.
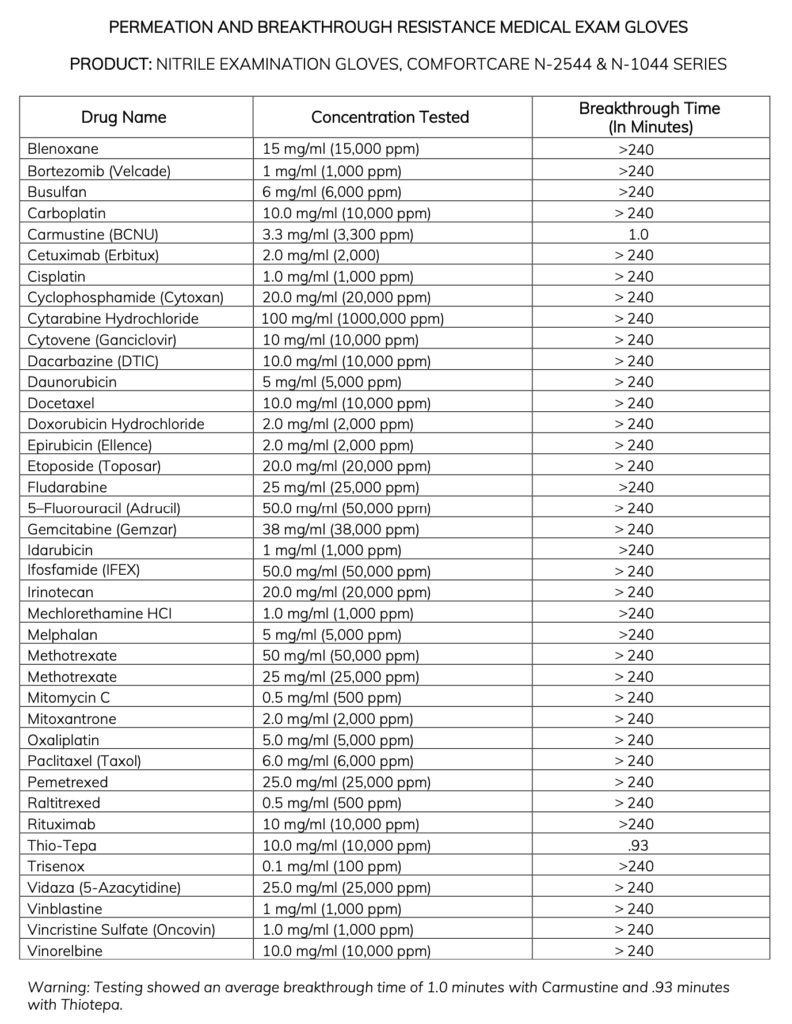
Chemo Testing Report N-2544 Comfortcare Premier 4.0:
Subj.: Chemotherapy Drug Permeation Resistance Outcomes
Drugs Tested for Representation
The chemotherapy gloves tested using the ASTM D6978 method encompass various classes of clinical drugs. Having representation from each class is crucial, as it would be impractical to test against every chemotherapy drug available. The ASTM testing methods mandate that at least nine currently used drugs be tested.
The chosen clinical drugs must comprise a minimum of the following seven drugs:
- Carmustine
- Cyclophosphamide
- Doxorubicin HCI (Adriamycin)
- Etoposide
- Fluorouracil (Adrucil)
- Paclitaxel (Taxol)
- Thio-Tepa
Two additional clinical drugs can be selected from a list supplied by ASTM in the testing method. These clinical drugs are procured from pharmaceutical manufacturers or authorized distributors. Each test drug is prepared using the recommended solvent from the manufacturer.
The drug solution is formulated with the suggested solvent and at the highest concentration to which a healthcare professional might be exposed during handling.
Breakthrough Time Results
The outcome for each chemo drug is presented as “breakthrough time,” which refers to the time required for the chemical to permeate from the glove’s outer surface to the inner surface. A longer breakthrough time indicates superior protection. To guarantee sufficient protection, the breakthrough time should exceed the glove wear time.